Fiona Kolbinger receives 2026 Jung Career Advancement Award for Medical Research
AI in Intensive Care
Two new publications provide an overview of
Artificial intelligence (AI) has entered intensive care medicine – but mostly in a limited, task-specific form. Two publications in npj Digital Medicine, led by Dr. Oscar Freyer together with Prof. Stephen Gilbert from EKFZ for Digital Health at TUD, provide a systematic overview of AI-enabled medical devices for intensive care units (ICUs) and examine how the field may evolve.

Current landscape: AI applications already in use in intensive care
In a comprehensive analysis, the researchers identified 36 AI-based medical devices authorized in the EU and the US for ICU use. This number is considerably higher than previously reported. However, most systems are narrowly focused and designed to support risk prediction, for example for sepsis, overall declining health status of a patient, or problems with blood pressure and circulation (hemodynamic instability). These systems typically rely on vital signs or electronic health record data rather than medical imaging.
The researchers applied a multi-method search, as no single database – especially in Europe – provides a complete overview of AI-based medical devices. Their approach combined database searches with a structured web search, followed by manual validation using regulatory documents and manufacturer information. Each device was characterized along regulatory and clinical/technical dimensions using an extended taxonomy.
Freyer O, Buch S, Bassily-Marcus A, Zenker S, Pickering BW, Ostermann M, Schönfelder A, Gilbert S. The landscape of artificial intelligence-enabled medical devices in the EU and the US intended for intensive care units, npj Digital Medicine, 2026. https://doi.org/10.1038/s41746-026-02609-2
From narrow tools to complex AI systems: the need for new regulatory approaches
The companion Perspective article outlines how AI in intensive care could develop further. The authors describe a transition from today’s single-purpose tools to more complex, agentic systems that could coordinate multiple data sources, devices, and patients simultaneously. Current regulatory frameworks, however, are built around clearly defined individual products and are not designed for such systems.
To structure this development, the research team proposes a five-paradigm framework based on scope and scale. It ranges from narrow systems affecting individual patients to more advanced systems that operate across multiple tasks and patients. The most advanced stage describes orchestrating, agentic AI systems that act at unit level. Such systems challenge existing regulatory approaches, as they no longer fit the model of single products with clearly defined purposes. The classification is based on the identified available devices, a structured literature review on AI applications in critical care, and a regulatory analysis of the EU Medical Device Regulation, the EU AI Act, the UK MDR, and the US FDA framework. The authors conclude that the emergence of generative and agentic AI systems presents new opportunities but also complex regulatory challenges, particularly concerning adaptability, autonomy, and system-level integration.
Overall, the work highlights two central gaps: limited transparency on approved AI medical devices in Europe, and a mismatch between existing regulation and emerging agentic, generative AI approaches. Addressing both will be essential for the safe and effective use of AI in critical care in the future.
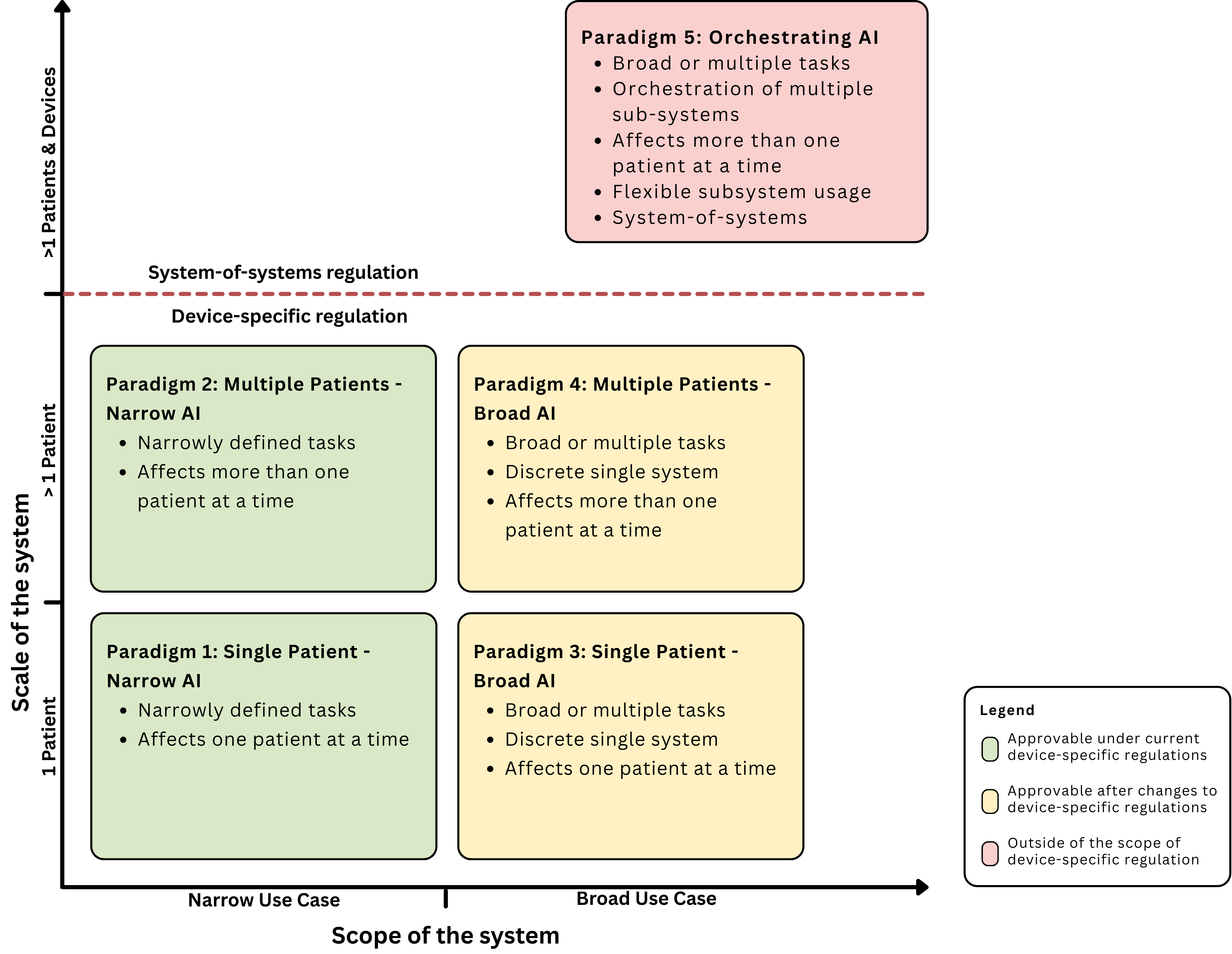
Freyer O, Mathias R, Muti HS, Orlovsky H, Buch S, Ostermann M, Schönfelder A, Poncette AS, Bassily-Marcus A, Gilbert S. The regulation of artificial intelligence in intensive care units: from narrow tools to generalist systems, npj Digital Medicine, 2026. https://doi.org/10.1038/s41746-026-02535-3
More News
Conductive Hydrogel Enables Electrical and Biochemical Signal Control





