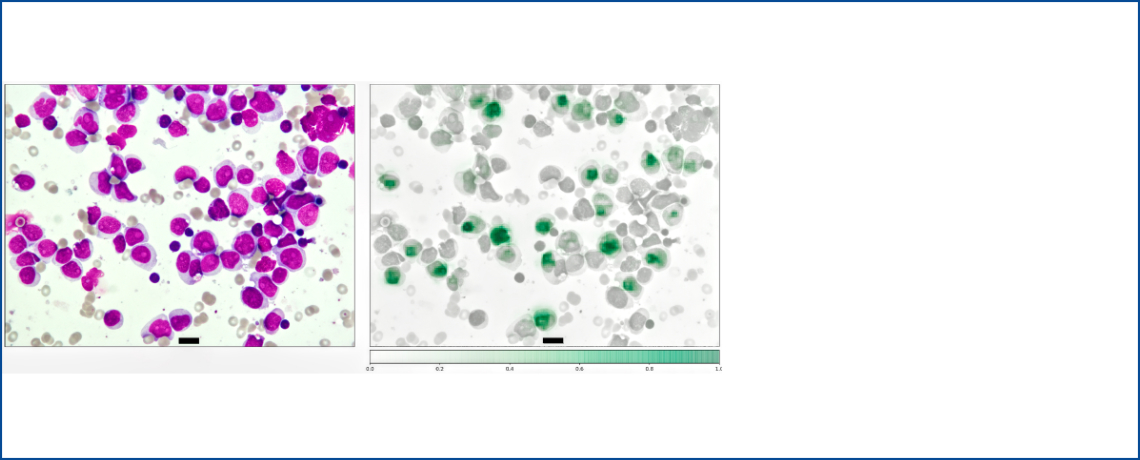
Machine Learning for Advanced Integrated Diagnostics of Myeloid Neoplasms
Medical Need
The ever-increasing complexity of individual patient data in hematology and oncology heralds an era of dynamic data-driven systems for personalized tumor diagnostics and patient management. As both cure rates and long-term survival for many malignant entities, especially myeloid leukemias, are still largely unsatisfactory, the development and clinical integration of novel methodologies in diagnostics, risk stratification and patient care are warranted. Machine learning and deep learning excel over conventional hypothesis-driven models as they are better suited to keep up with the rapid pace of advancements in our understanding of tumor biology and may thus facilitate the adoption of personalized care in clinical routine via fast and robust decision support algorithms.
With MAINFRAME, we aim to advance our pre-existing AI prototypes in order to provide a seamless and automated decision support pipeline for image-based bone marrow diagnostics as well as unveil insights into the molecular biology of myeloid neoplasms via big data analysis of large-scale gene banks.
Machine Learning, Artificial Intelligence, Computer Vision, Big Data, Acute Myeloid Leukemia, AML, Myelodysplastic Syndromes, MDS, Hematology, Oncology, Bone Marrow, Genetics
Project team members
Clinician
University Hospital Carl Gustav Carus, Department of Internal Medicine I
Project team members
High-tech
TU Dresden, Institute of Software and Multimedia Technology
Abstract
With the advent of Machine Learning (ML) in medicine, hypothesis-driven approaches of data analysis and manual analog microscopy for research and diagnostic purposes in hematology seem outdated, tedious and often generate irreproducible results. With its capability to make sense of large and heterogenous data sets, ML is well suited to both provide insights into disease biology using a data-driven approach and automatize bone marrow image analysis via computer vision. MAINFRAME is a series of interconnected ML projects building on our existing prototypes for the analysis of complex clinical and genetic data from cancer patients as well as bone marrow image data. Thereby, we aim to provide a clinically relevant decision support tool for the automated analysis of bone marrow smears both in initial diagnosis as well as during disease monitoring for hematological neoplasms. Further, using large knowledge banks with multimodal data from cancer patients, we aim to discover genetic drivers, risk profiles and ultimately a way to personalized therapy in myeloid malignancies such as acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS).

